If you have worked with iPSC-derived hepatocytes over the past decade, you may have used DefiniGEN cells. DefiniGEN was one of the first companies to commercialise human iPSC-derived hepatocytes at scale, and its Opti-HEP product became a reference model for a generation of drug discovery and safety research programmes.
In July 2025, DefiniGEN was acquired by Phenaros, and from that merger pixlbio was formed. This post explains what happened, what changed, and what it means for researchers who have used DefiniGEN products or are considering iPSC-derived hepatocytes for the first time.
The History
DefiniGEN was founded in Cambridge in 2012, spun out from the laboratory of Ludovic Vallier at the Wellcome Sanger Institute. Its founding scientific premise was that iPSC-derived hepatocytes could be differentiated to a level of maturity that made them genuinely useful for drug metabolism and toxicology applications, not just for developmental biology research.
Over the following decade, DefiniGEN developed and commercialised Opti-HEP, an iPSC-derived hepatocyte product with validated CYP450 activity, albumin secretion, and urea synthesis. Opti-HEP was used by pharmaceutical companies, academic laboratories, and contract research organisations for hepatotoxicity screening, drug metabolism studies, and liver disease modelling. Several publications using DefiniGEN hepatocytes in peer-reviewed studies contributed to the scientific characterisation of what iPSC-derived hepatocytes could and could not do.
Phenaros, founded in Uppsala and led by Christopher Trummer with scientific co-founders Ola Spjuth, Jordi Carreras-Puigvert and Morgan Ekmefjord, had built a complementary capability: high-throughput Cell Painting phenomics, AI-driven image analysis, and predictive modelling applied to biological data from human cell models. Where DefiniGEN's strength was in the biology of the cell, Phenaros's strength was in extracting predictive information from that biology at scale.
The merger combined these capabilities into a single integrated platform: human-relevant iPSC-derived cell models generating phenomics data interpreted by AI-driven analytics. That integrated platform is pixlbio.
What Changed and What Did Not
The product names changed. Opti-HEP is now pixHep. The hepatic stellate cell product is now pixStellate. The intestinal organoid product is now pixIO. The Cell Painting and phenomics service is pixCellPaint, and the end-to-end research service is pixCellServices.
The underlying biology did not change. The differentiation protocols that generated Opti-HEP were developed over more than a decade by the DefiniGEN scientific team, led by Dr Nikolaos Nikolaou, who is now Global Headof Cell Biology at pixlbio and continues to lead the development and optimisation of the hepatocyte differentiation platform. The institutional knowledge, the validated protocols, the QC frameworks, and the scientific rigour that characterised DefiniGEN's cell products are part of pixlbio.
The team did not change significantly. The core scientists who developed and validated DefiniGEN's cell products are the same scientists who validate and supply pixHep today.
What pixHep Is Relative to Opti-HEP
pixHep is a continuation and extension of the Opti-HEP product line, not a replacement developed from scratch.
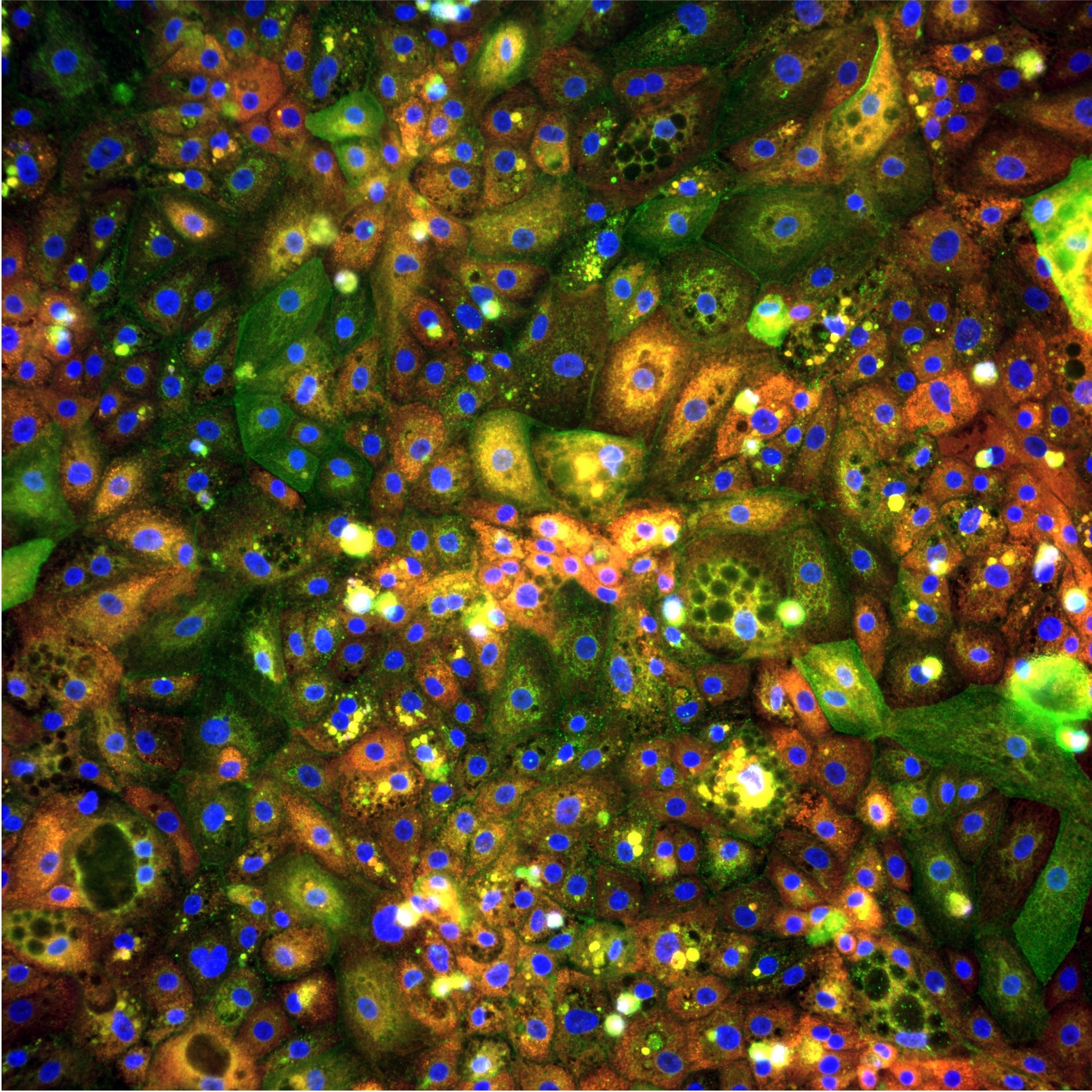
The core differentiation protocol is the same three-step protocol described in the 2024 bioRxiv preprint by Nikolaou et al., which characterised the generation of iPSC-derived hepatocytes with CYP450 expressionand functional performance comparable to primary human hepatocytes across multiple functional endpoints. The preprint presented data showing CYP3A4 inducibility, albumin secretion, urea synthesis, de novo gluconeogenesis, ASGR1-mediated GalNAc-siRNA uptake, and accurate DILI prediction for a panel of compounds with known hepatotoxic liability.
What has been extended relative to DefiniGEN's original product offering:
The disease model portfolio has been substantially expanded. pixlbio now offers MASLD models with PNPLA3 I148M and TM6SF2 E167K CRISPR-engineered risk variants alongside multiple wild-type donor backgrounds. Disease models for A1ATD, PFIC2, UCD, and other inherited metabolic liver diseases are available as part of the pixCells portfolio.
The analytical infrastructure has been substantially upgraded. Where DefiniGEN supplied cells with QC data and left downstream analysis to the customer, pixlbio supplies cells as part of an integrated service that includes Cell Painting phenomics, AI-driven feature extraction, and structured data delivery. Customers can receive cells only, or cellscombined with full analytical services, depending on their internal capabilities.
The multi-donor panel has been extended. pixHep is now available from multiple validated wild-type donor backgrounds with documented genetic diversity, supporting the multi-donor experimental design philosophy described in our previous post on donor variability.
What This Means If You Have Used DefiniGEN
If you have published data using DefiniGEN Opti-HEP cells and want to continue using the same biological system, pixHep provides continuity. The differentiation protocol is the same, the QC criteria are equivalent, and the same scientific team maintains the platform.
Historical comparability is an important consideration forlaboratories that have built validated assay platforms around DefiniGEN cells. We understand this and have specifically maintained protocol continuity to preserve the ability to compare new data with historical DefiniGEN-derived results.
If you want to access capabilities that were not available through DefiniGEN, including the expanded disease model portfolio, the Cell Painting phenomics service, and the multi-donor experimental design support, these are now available through pixlbio as extensions of the core biological platform.
What This Means If You Are Evaluating iPSC Hepatocytes for the First Time
If you have not previously worked with DefiniGEN and are evaluating iPSC-derived hepatocytes for the first time, the DefiniGEN history is relevant as evidence of track record.
Opti-HEP was used successfully for drug metabolism studies, hepatotoxicity screening, and liver disease modelling by pharmaceutical companies including AstraZeneca, GSK, Deep Genomics, and others over many years. The scientific publications, the customer relationships, and the operational experience built over that period are part of the institutional foundation of pixlbio.
The merged company combines that track record in cellbiology with the AI phenomics and predictive modelling capabilities developedat Phenaros, creating a platform that neither predecessor company could have offered independently.
The Scientific Rationale for the Merger
The combination of high-quality human cell models with scalable, AI-driven phenomics was not accidental. It reflects a specific scientific conviction: that the value of human-relevant cell models in drugdiscovery is fully realised only when the data those models generate is interpreted by analytical systems capable of extracting its full information content.
A high-quality iPSC-derived hepatocyte that is analysed by aviability assay alone is an expensive way to generate a binary live-dead signal. The same hepatocyte, analysed by Cell Painting morphological profiling and AI-driven feature extraction, generates a multi-dimensional phenotypic signature that can be used for mechanism of action classification, toxicity prediction, disease phenotype characterisation, and donor-stratified response mapping simultaneously.
That is the integrated value proposition of pixlbio, and it is the reason the merger made scientific as well as commercial sense.
Conclusion
- Huch M et al. (2015). Long-term culture of genome-stable bipotent stem cells from adult human liver. Cell
- Vallier L et al. (2009). Signaling pathways controlling pluripotency and early cell fate decisions of human induced pluripotent stem cells. Stem Cells
- Nikolaou N et al. (2024). Development of optimised human iPSC-derived hepatocytes with improved liver function for in vitro metabolic disease modelling and toxicity studies. bioRxiv
- Godoy P et al. (2013). Recent advances in 2D and 3D in vitro systems using primary hepatocytes, alternative hepatocyte sources and non-parenchymal liver cells. Archives of Toxicology
- Bray MA et al. (2016). Cell Painting, a high-content image-based assay for morphological profiling. Nature Protocols