Pixl Bio (formerly DefiniGEN) is one of eight commercial liver microphysiological system (MPS) developers selected for the most rigorous cross-platform evaluation of liver MPS conducted to date — a public–private consortium co-led by FDA-CDER and the 3Rs Collaborative, with independent data governance by NIH-NIEHS/NICEATM. The project's Letter of Intent was accepted into the FDA's ISTAND qualification program in January 2026.
Presented at the Society of Toxicology 65th Annual Meeting and ToxExpo · March 22–25, 2026 · San Diego, CA · Abstract 3384 / Poster G527
Why This Study Matters for Liver MPS
Drug-induced liver injury (DILI) is one of the most consequential failure modes in drug development. It is a leading cause of late-stage attrition and post-market regulatory action — a problem that existing preclinical models have consistently failed to solve. Conventional two-dimensional hepatocyte cultures and animal studies have a well-documented record of missing human hepatotoxic signals, leaving developers exposed to costly clinical surprises.
Liver microphysiological systems offer a fundamentally more human-relevant alternative. By reconstructing the cellular microenvironment of the liver in three dimensions, these systems can detect injury signals that flat cultures cannot. Despite this promise, liver MPS adoption in formal regulatory submissions has been slow — held back by the absence of cross-platform performance data and a clearly defined regulatory context of use (CoU).
This study, presented at the Society of Toxicology Annual Meeting in March 2026, was designed specifically to close that gap.
About the Project
The study was established under a formal public–private partnership, with FDA-CDER providing project leadership and guidance, the 3Rs Collaborative handling project coordination and dissemination, NIH-NIEHS/NICEATM providing compound selection, blinding, independent data analysis and technical scientific support, and the Critical Path Institute contributing context of use and qualification expertise.
The project's scope was deliberately narrow. Before any testing began, stakeholders agreed on a specific, clinically anchored context of use:
Use of a liver MPS as a retrospective analytical tool to evaluate elevated liver enzymes (ALT/AST) observed in early-phase clinical trials, providing weight-of-evidence to inform continuation or modification of dosing.
This framing is significant. The study is explicitly platform-agnostic and aligned with the FDA's ISTAND qualification pathway, which emphasises category-level evidence generation rather than endorsement of individual products. The consortium's Letter of Intent for this context of use was accepted into ISTAND in January 2026 — a first-of-its-kind milestone for cross-platform liver MPS qualification.
The three-stage ISTAND process — LOI, Qualification Plan, Full Qualification Package — provides a clear pathway toward regulatory acceptance. Once qualified, a drug development tool can be included in IND, NDA, or BLA applications without requiring FDA to re-evaluate its suitability for the stated CoU.
Pixl Bio's Role
Pixl Bio (operating as DefiniGEN at the time of the study) was one of eight selected commercial liver MPS developers invited to participate. Our platform — built on proprietary iPSC-to-hepatocyte differentiation technology — provided data across five endpoints against the full blinded compound set:
- ALT (clinical liver injury marker; high clinical DILI relevance)
- AST (clinical liver injury marker; high clinical DILI relevance)
- CYP3A4 activity (Phase I metabolic function; high clinical relevance)
- Albumin (hepatocyte function marker)
- ATP
The combination of hepatocellular injury markers (ALT, AST) alongside functional indicators (CYP3A4, albumin, ATP) positioned Pixl Bio as a strong contributor, covering endpoints across all clinically relevant tiers — from high-relevance injury markers (ALT, AST, CYP3A4) to hepatocyte function (albumin) and cellular bioenergetics (ATP) — directly mirroring the clinical endpoints that prompt regulatory concern in early-phase trials.
Pixl Bio is now the active brand; DefiniGEN is the legacy name under which this scientific work was conducted.
The Consortium
The project spans five stakeholder categories, governed by independent data management. NIH-NIEHS/NICEATM was responsible for compound selection, blinding, and all statistical analysis — insulating the outcome from commercial influence.
Study Design
All eight platforms were tested against the same eight blinded compounds using harmonised principles adapted to each platform's design. The experimental strategy included:
- 8 drugs with known clinical truth: 4 associated with high DILI risk and 4 structurally related low-risk controls
- Exposure levels anchored to multiples of human Cmax
- Repeated dosing for 3–14 days, depending on platform capabilities
- Harmonised solvent conditions (0.2% DMSO)
- GLP-like documentation and quality control encouraged across all sites
The study deliberately did not require identical protocols across platforms — a design choice that enabled genuinely diverse technologies to participate on equal footing, including iPSC-based systems like Pixl Bio's. This approach is consistent with the principles described by Ewart et al. in their 2022 performance assessment of human Liver-Chip models, where rigorous compound-blinding and clinically anchored endpoints were central to establishing regulatory-grade evidence for DILI prediction. Independent data management and statistical evaluation was handled entirely by NICEATM, with no platform seeing another's data during the blinded phase.
Study at a Glance
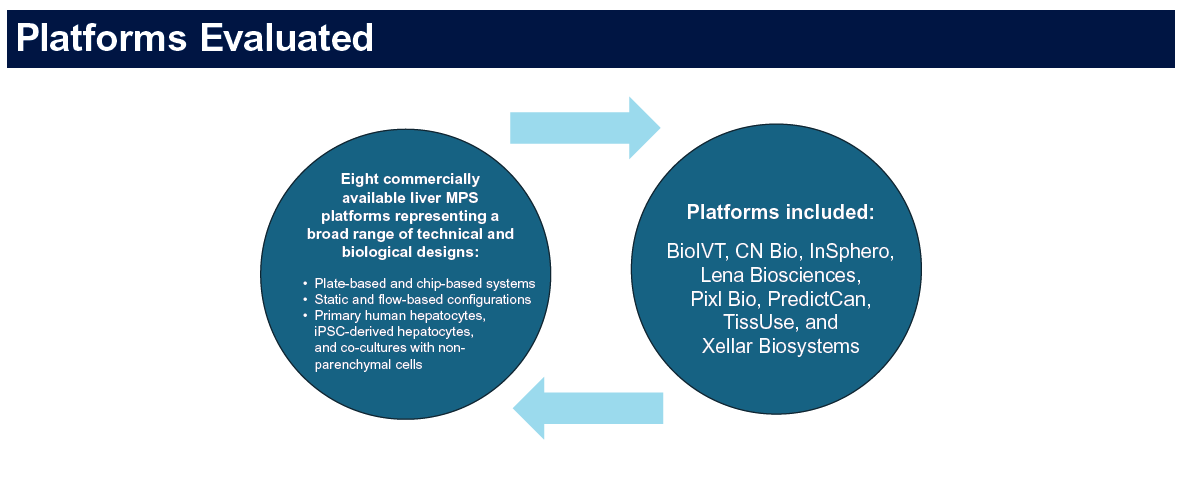
- 8 commercial liver MPS platforms evaluated, including Pixl Bio
- 8 blinded test compounds — 4 high DILI risk, 4 low-risk structural analogues
- 5 endpoints provided by Pixl Bio: ALT, AST, CYP3A4, albumin, ATP
- 2026 target year for results paper and ISTAND qualification plan submission
Project Timeline
Preliminary Results
Analysis is ongoing, but initial findings are encouraging. Most platforms successfully distinguished the safer compound within each of the four drug analogue pairs. Most technologies correctly identified high versus low DILI concern, with some platforms demonstrating more granular categorisation — high, moderate, low, and no DILI concern — within the compound set.
Follow-up transcriptomic and LC-MS profiling is planned to provide mechanistic depth, with full results expected for publication in 2026.
Why This Is Significant for Pixl Bio
Participation in this study is not merely a validation exercise. It is a direct contribution to the regulatory evidence base that will shape how liver MPS technologies are used in drug development globally. By generating blinded, CoU-anchored performance data alongside seven other platforms, Pixl Bio has helped build the category-level confidence that regulators need to act.
The study also demonstrates how Pixl Bio's liver MPS platform is positioned: not as an incremental screening tool, but as a human-relevant system capable of informing critical go/no-go decisions in early clinical development. The ability to measure ALT, AST, and CYP3A4 in the same system — mirroring the clinical endpoints that prompt regulatory concern — is precisely what makes the platform relevant to this CoU.
As the field moves toward formal ISTAND qualification, Pixl Bio will continue to contribute data, expertise, and scientific leadership to the regulatory science of liver MPS qualification. Our work on disease models including MASLD, A1ATD, and PFIC2 places us at the intersection of human-relevant biology and regulatory-grade evidence generation.
Key Lessons from the Study
A narrow CoU is essential. Defining a specific, clinically relevant question before testing was critical to both feasibility and regulatory credibility. The NICEATM/ICCVAM framework for NAM validation is clear on this: validation should establish fitness for a specific purpose, not blanket capability.
Harmonisation of principles, not protocols. Requiring identical workflows would have excluded most platforms. Harmonising scientific principles — Cmax anchoring, dosing duration, endpoint families — while permitting platform-specific execution enabled genuine diversity, including iPSC-based platforms like Pixl Bio's.
Independent governance is non-negotiable. NICEATM's role as independent data steward was structurally critical to the study's regulatory credibility. No platform saw another's data during the blinded phase.
Legal complexity needs early attention. Material transfer agreements and research collaboration agreements were a significant time constraint. Future multi-platform studies should negotiate standard templates before compounds are even selected.
For more information about Pixl Bio's liver MPS platform and its application to regulatory toxicology, DILI, and human disease modelling, contact our team.